Dr Surjit Singh Bhatti, (Retired) Professor & Head Physics, and Dean, Faculty of Sciences, Guru Nanak Dev University, Amritsar.
The coveted 2019 Nobel Prize for Chemistry was awarded by The Royal Swedish Academy of Sciences jointly to Dr John B. Goodenough and Dr M. Stanley Whittingham (both of USA) and Dr Akira Yoshino (of Japan),
“for the development of lithium-ion batteries.”
These are powerful rechargeable and very compact batteries, being used in mobile phones, computers and many other portable electronic devices. Since these batteries can store large amount of electrical charge, from both conventional as also the renewable sources of energy, they are used in communication networks as well as electric vehicles and aircrafts.
Interestingly, before the announcement was made, some Scientific Organizations had asked a large number of scientists to vote for who they thought had the best chance to get the 2019 Nobel prize for chemistry. Nine names were suggested, based on predictions of a panel of reputed researchers. Overwhelming majority of the votes went to the 97 years old Dr John B. Goodenough of Texas, USA, and two others who helped him.
Dr John B. Goodenough was born in 1922 in Germany. He got his Ph.D. in 1952 from the University of Chicago, USA. He is Chair of the Department of Engineering, University of Texas at Austin, USA.
Dr M. Stanley Whittingham was born in 1941 in the UK. He got his Ph.D. in 1968 from Oxford University, UK. Later, he became a Distinguished Professor at Binghamton University, State University of New York, USA.
Dr Akira Yoshino was born in 1948 in Japan. He got his Ph.D. in 2005 from Osaka University, Japan. He is Honorary Fellow at Asahi Kasei Corporation, Tokyo and Professor at Meijo University, Nagoya, Japan.
Early Batteries: In a cell, chemical energy is converted to electrical energy. Recharging reverses this process. The earliest Lead-Acid batteries have six cells, which produce 12.6 Volts of Energy. Each cell has two Lead (Pb) plates, the positive electrode is covered with a paste of Lead dioxide (PbO2) and the negative electrode is made of sponge Lead. During discharge, at the anode, electrons and Lead sulfate (PbSO4) are produced due to reaction with the sulfuric acid in the electrolyte. The lead oxide is reduced to PbSO4 at the cathode. In these batteries, the disadvantage is that Lead(Pb) gives low specific energy (energy per unit mass or energy density) due to its high atomic weight.
Next came the Nickel-Cadmium (Ni-Cd) battery with Nickel oxide hydroxide (Ni O-OH) and metallic Cadmium (Cd) as electrodes and Potassium hydroxide (KOH) as the electrolyte. Its working is similar to that of the Lead-Acid battery.
The Li-ion battery, likewise, is composed of two electrodes, connected to an electric circuit and separated by an electrolyte. When the battery drives the electric current, an oxidation process takes place at the anode and reduction takes place at the cathode. Electrons (e–) move through the outer circuit while the Li-ions (Li+) move through the battery; providing electrical energy. Since Lithium has a much smaller atomic weight (compared with Lead), its specific energy is very high. This gives a higher cell voltage which depends on the potential difference of the electrodes.
In 1976, Whittingham discovered that a cathode made of Titanium disulphide (TiS2), in a Li-ion battery, can store in its spaces a much larger number of ions released by the metallic Lithium anode. On charging, these ions flow back again. In 1980, Goodenough showed that Cobalt oxide (CoO2) produces a higher voltage, leading to even more powerful batteries. In 1985, Akira Yoshino used a Carbon (C) material in place of the reactive Lithium for the anode. Lithium ions could be interspersed or intercalated more easily in the molecular layers in petroleum coke, which is a form of Carbon. When the battery is being used, the ions flow between the electrodes without reacting with their surroundings, giving it a longer life.
This battery, with a much better performance, became commercial in 1991. Goodenough later replaced the Cobalt oxide cathode with Iron or Manganese phosphate, as these have longer life and less risks. They are also more environment friendly.
At the positive electrode, (reversible) reaction in Li-doped Cobalt oxide is:
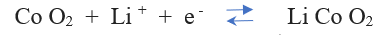
At the negative electrode, (the reversible) reaction for the Carbon ( C6 ) is:
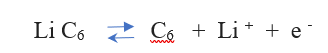
The reaction from left to right is discharging, from right to left is charging.
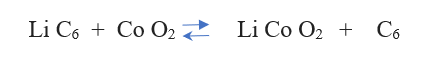
Solid Electrolytes (ceramics) are being used in advanced Li-ion batteries these days. Mostly Li metal oxides are used which allow Li-ion transport through the solid more readily. There is no risk of leaks, which is a serious safety concern with the Lead-Acid and Ni-Cd batteries having liquid electrolytes. It is a step towards fossil-free, clean energy.
Popularity of Lithium ion batteries is basically due to the advantages of their high energy density, low cost of maintenance, low self-discharge, quick charging, compactness and longevity. Moreover, very pure (and expensive) Lithium is not required in these batteries.
Safety considerations are important as their thermal stability is relatively low. High temperatures may cause fire and even explosion. To overcome these risks, one has to prevent over-heating (above 800C), over-charging, external short-circuiting and Lithium dendrite formation.